COVID-19 Drug Delivery Devices Market Is Likely To Witness A Healthy CAGR Growth of USD 2.15 Billion By 2027 | Grand View Research, Inc.
According to a report, “COVID-19 Drug Delivery Devices Market Size, Share & Trends Analysis Report By Product (Prefilled Syringe, Patches), By Route of Administration, By Distribution Channel, By Region, And Segment Forecasts, 2022 – 2027”, published by Grand View Research, Inc., The global COVID-19 drug delivery devices market is anticipated to be worth USD 2.15 billion by the year 2027, advancing at a healthy CAGR of 8.1% during this period, according to a recent GVR report. The spread of the COVID-19 pandemic across the world since the beginning of 2020 has been swift, with countries, both developed and developing, reeling from its impact. With more than 15 million confirmed cases and more than 610,000 deaths globally, the pandemic is expected to sustain its impact over the coming months. The outbreak has seen an unprecedented surge in research and development of various drugs and vaccines, with many established organizations entering into partnerships to develop an effective combatant to coronavirus.
The drug development phase began almost as soon as the pandemic started its global spread from China; however, there are still vast improvements to be made in this regards, with many of them still in the trial phase, which would ensure the growth of the market for COVID-19 drug delivery devices over the coming year. The US FDA has fast-tracked the drug approval phase for manufacturers, with the country being the leading in terms of both the number of confirmed cases as well as deaths, which has necessitated the distribution of an effective drug to the affected population at the earliest.
The market is also expected to witness sharp growth in the coming months, on account of initiatives and partnerships undertaken by organizations and economies to bring vaccines for people at the global level. The WHO set up a Research and Development (R&D) Blueprint in April 2020, with the aim to speed up development of diagnostics, vaccines, and therapeutics for the pandemic. Additionally, a number of economies entered into partnerships and alliances for exchanging knowledge and resources to come up with an effective solution. An example is the Inclusive Vaccine Alliance, formed by France, Germany, Italy, and the Netherlands, which has partnered with AstraZeneca for the supply 300 to 400 million doses of vaccine in Europe post-approval. Additionally, India, where the pandemic has shown no signs of stopping, has been granted funds to support vaccine development from a number of sources, such as the DBT-BIRAC consortium, while relaxations have also been allowed on previously stringent norms to speed up vaccine development.
“Would you Like/Try a Sample Report” Click the link below: https://www.grandviewresearch.com/industry-analysis/covid-19-drug-delivery-devices-market/request/rs5
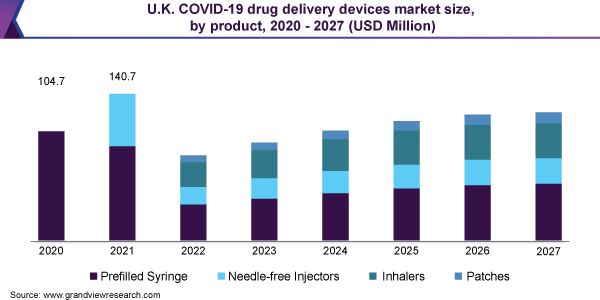
The global COVID-19 drug delivery devices market has been segmented on the basis of product, route of administration and distribution channel. In terms of product, the market is classified into prefilled syringe, needle-free injectors, inhalers and patch. The prefilled syringe segment is expected to lead the market in the near future, as their bulk production will make them one of the first products to be approved in coming months. Needle-free injectors are also expected to show substantial growth in this period, as they eliminate the occurrence of needlestick injury (NSI), which is a cause for transmission of fatal diseases such as HIV and hepatitis. The CDC has stated that NSIs cost an estimated USD 3,042 per victim annually in U.S.
Based on the administration route, the market has been segmented into parenteral, nasal and dermal. Out of these, the parenteral segment is expected to account for a substantial market share in the future, owing to its affordability and accessibility. Till very recently, 10 potential COVID-19 vaccine candidates have entered the clinical trials, out of which majority are to be administered through the parenteral route. The distribution channels for the market include hospital, retail and online pharmacies, out of which hospital pharmacies are expected to assert dominance in the coming years. As recently as June 2020, the National Association of Chain Drug Stores (NACDS) requested the US government to depend on pharmacies for rapid COVID-19 vaccine deployments, which is expected to boost the segment growth.
Trends and Developments:
- A number of drugs such as the combination of lopinavir and ritonavir, which are used in cases of HIV, are currently being evaluated for the treatment of COVID-19; however, their uncontrolled dosage can cause grave issues such as diarrhea, nausea and liver damage in patients. The development of a controlled release formulation is expected to reduce the burden on the liver by 81%. Thus, similar developments in this area can result in optimum drug release in the target patients.
- A combination of concepts by the Chalmers University of Technology, Sweden, the University of Gothenburg, and AstraZeneca, among others, has resulted in the investigation of a unique nasal spray mRNA vaccine to combat COVID-19. Further research is expected to show the efficacy of this method for combating the pandemic.
- PharmaJet, which specializes in developing needle-free injections, teamed up with Abnova Corporation, a Taiwan based antibody manufacturer, in May 2020, for the development of a needle-free injection system technology to deliver a messenger RNA (mRNA) vaccine against coronavirus.
- Vesicular drug-delivery systems are being employed as co-delivery tools for personalized theranostics that combine diagnostic, prognostic therapeutic and image-guided therapeutic effects.
Have Any Query? Ask Our Experts for More Details on Report: https://www.grandviewresearch.com/inquiry/450617/ibb
Route of Administration Insights
The parenteral segment is expected to capture more than 60.0% share of the overall revenue in 2021 and is anticipated to maintain its lead over the forecast period. Novel biologics such as RNA and DNA-based vaccines, recombinant nanoparticles, and monoclonal antibodies are currently being explored in clinical trials. Such biologics can only be administered intravenously as they are degraded when given orally. Currently, about 10 potential COVID-19 vaccine candidates have entered the clinical trials, most of which are to be administered via a parenteral route of administration. On 1st June 2020, Moderna initiated a Phase II clinical trial for mRNA-1273, a COVID-19 vaccine candidate. The company partnered with the Vaccine Research Center (VRC) at the National Institute of Allergy and Infectious Diseases (NIAID) for vaccine development.
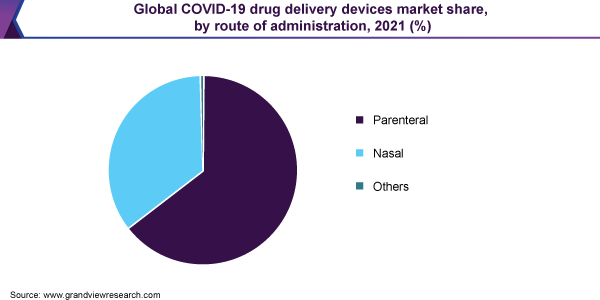
The nasal segment is anticipated to witness significant growth over the forecast period as the drug and vaccine manufacturers are focused on developing vaccines, which would reduce storage and administration costs. Thus, the innovative approach helps in eliminating disease transmission and needle stick injuries, thus saving medical costs in the already burdened healthcare settings.
COVID-19 Drug Delivery Devices Market Segmentation
Grand View Research has segmented the global COVID-19 drug delivery devices market based on product, route of administration, distribution channel, and region:
COVID-19 Drug Delivery Devices Product Outlook (Revenue, USD Million, 2020 – 2027)
-
Prefilled Syringe
-
Needle-free Injectors
-
Inhalers
-
Patch
COVID-19 Drug Delivery Devices Route of Administration Outlook (Revenue, USD Million, 2020 – 2027)
-
Parenteral
-
Nasal
-
Dermal
COVID-19 Drug Delivery Devices Distribution Channel Outlook (Revenue, USD Million, 2020 – 2027)
-
Hospital Pharmacies
-
Retail Pharmacies
-
Online Pharmacies
COVID-19 Drug Delivery Devices Regional Outlook (Revenue, USD Million, 2020 – 2027)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
U.K.
-
Germany
-
France
-
Italy
-
Spain
-
-
Asia Pacific
-
Japan
-
China
-
India
-
South Korea
-
Australia
-
-
Latin America
-
Brazil
-
Mexico
-
Argentina
-
Colombia
-
-
Middle East & Africa
-
South Africa
-
Saudi Arabia
-
UAE
-
List of Key Players of COVID-19 Drug Delivery Devices Market
-
AstraZeneca
-
Johnson & Johnson
-
Serum Institute of India
-
GlaxoSmithKline plc
-
Moderna
-
Bharat Biotech
-
Pfizer
-
PharmaJet
-
Novawax, Inc.
Request for Customization : https://www.grandviewresearch.com/request-for-customization/450617/rfc1
Explore the BI enabled intuitive market research database, Navigate with Grand View Compass, by Grand View Research, Inc.
About Grand View Research
Grand View Research provides syndicated as well as customized research reports and consulting services on 46 industries across 25 major countries worldwide. This U.S.-based market research and consulting company is registered in California and headquartered in San Francisco. Comprising over 425 analysts and consultants, the company adds 1200+ market research reports to its extensive database each year. Supported by an interactive market intelligence platform, the team at Grand View Research guides Fortune 500 companies and prominent academic institutes in comprehending the global and regional business environment and carefully identifying future opportunities.
Media Contact
Company Name: Grand View Research, Inc.
Contact Person: Sherry James, Corporate Sales Specialist – U.S.A.
Email: Send Email
Phone: 1-415-349-0058, Toll Free: 1-888-202-9519
Address:201, Spear Street, 1100
City: San Francisco
State: California
Country: United States
Website: https://www.grandviewresearch.com/industry-analysis/covid-19-drug-delivery-devices-market



